Solar keratoses of the scalp and a brisk reaction to topical ingenol mebutate
An elderly man with a history of sun-damaged skin experiences a painful reaction when solar keratoses of the scalp are treated with ingenol mebutate gel. What is this reaction and how should a patient be managed in this situation?
Case scenario
A 76-year-old man presented to his GP for a tender scalp. He had a long history of chronic solar damage, with multiple squamous cell carcinomas having been excised in the past. Apart from multiple areas of skin grafts, he continued to have extensive field changes to the remaining scalp. Sheets of small actinic keratoses could be found, as well as areas of thicker scaly skin changes.
The GP decided to use a ‘field treatment’ to treat the entire scalp, as targeting individual lesions over the scalp was impractical. Ingenol mebutate 0.015% gel was prescribed, to be applied for three days to the scalp.
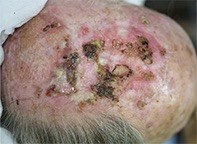
The day after the second application of ingenol mebutate, the patient woke with intense pain, and swelling and blistering of the scalp. Examination revealed areas of redness, scaling, erosions and accumulated pus (Figure). In addition, parts of his partial thickness skin grafts had undergone necrosis, with exposure of the underlying cranium.
- Could the reaction indicate hypersensitivity? Is it likely to be an infection?
- How should the patient be managed?
Commentary
Ingenol mebutate is the newest treatment available in the Australian market for the treatment of actinic keratoses.1 It is intended as a field treatment, to be applied to an area of 25 cm2. In practice, most patients are advised to treat a discrete area, such as the forehead, either cheek or the central scalp. There are two preparations of ingenol mebutate available: a three-tube pack at 0.015% dosage strength indicated for face and scalp, and a two-tube pack at 0.05% dosage strength indicated for the rest of the body. Each tube contains enough gel for one application for one day. The published efficacy of topical ingenol mebutate gel is comparable to other field treatments (e.g. 5-fluorouracil cream or photodynamic therapy with methyl aminolevulinate), being at least a 50% reduction in countable actinic keratoses.2,3
Most patients have a very mild skin reaction to topical ingenol mebutate. Indeed, the competitive advantage of the medication is a gentle skin reaction together with a short course of treatment (just one application per day over two or three days). However, during the clinical trials, a very small number of subjects experienced an exaggerated inflammatory response with widespread erythema, scale, erosions, and lakes of purulent discharge. Regardless of the degree of inflammation, all reactions settled within a two-week period with no long-term sequelae. The pus is sterile and represents inflammation rather than infection. The reaction is not a true hypersensitivity, and is a pattern peculiar to ingenol mebutate. In practice, however, the frequency of the severe reaction seems to be more frequent – up to 10% of patients react vigorously. There is no pre-existing feature that would consistently predict the reaction.
The treatments of choice for actinic field damage featuring actinic keratoses remain 5-fluorouracil and photodynamic therapy (both daylight and lamp-based). Patients who instead choose ingenol mebutate are after a quick treatment with a mild local skin reaction profile. However, they must be made aware that a significant minority of patients can have an exaggerated inflammatory reaction. Although not specified in the formal product information for ingenol mebutate, the use of a mid-potency to potent topical corticosteroid is useful in rapidly settling the reaction.
Outcome
Upon review by a dermatologist, the patient was advised to cease treatment and to not complete the third application of ingenol mebutate gel. Very gentle debridement was performed to remove the dried-on pus, and the area was treated with a mid-potency topical corticosteroid under wet dressings twice daily for the next five days. The reduction in pain was rapid and the inflammatory areas settled within two weeks.
The perforated skin grafts did not recover; the decision was made to treat these conservatively. Partial thickness skin grafts over the scalp can be rather delicate, and care needs to be taken when any treatment that creates direct trauma or vigorous inflammation is used on or near them. If there is only minimal subcutaneous tissue supporting the graft then necrosis is possible. MT