Varicella – more than just a rash
Often considered a benign childhood illness, varicella (chickenpox) can be associated with serious and life-threatening complications. GPs have an important role in providing varicella vaccinations and in identifying patients with varicella infection or exposure, especially high-risk individuals who require counselling, prophylaxis and possible hospital referral.
Varicella (also known as chickenpox) is a highly contagious viral illness caused by primary infection with varicella zoster virus (VZV). It is characterised by general malaise and fever followed by the eruption of a vesicular rash over the next three to five days.1-3 Reactivation of VZV causes herpes zoster (shingles), a disease most prevalent in people aged over 60 years.4
Chickenpox is commonly seen in general practice among unvaccinated individuals. Often considered a benign childhood illness, it can be associated with serious and life-threatening complications, including pneumonia and disseminated disease.
This article focuses on chickenpox in childhood. It summarises the role of varicella vaccination and management of individuals at high risk of serious complications, including postexposure prophylaxis and antiviral treatment of chickenpox.
Epidemiology
Chickenpox is common in children aged under 15 years, with more than 90% of the unvaccinated population in temperate climates developing clinical infection or subclinical seroconversion by adolescence.5,6 Before the introduction of universal vaccination, there were about 240,000 cases and 1500 hospitalisations each year from varicella in Australia.7
After the introduction of a single-dose varicella vaccine under the National Immunisation Program (NIP) in 2005, hospitalisations for chickenpox declined rapidly in all age groups under 40 years.2,7 The greatest reduction (84%) was in children aged 18 to 59 months. In 2016, there were 395 varicella-coded hospitalisations, of which 101 (26%) were in children aged under 15 years.1
Between 1997 and 2016, 132 deaths were recorded from chickenpox in Australia. Among these, 15 deaths were in children aged under 15 years, only two of which occurred after vaccination commenced.1 The rates of varicella-related hospitalisations and deaths in Australia before and after the introduction of varicella vaccine are shown in Figures 1a and b.1
Pathogenesis
VZV is a double-stranded DNA virus and member of the herpes virus family.8 VZV infection causes two distinct clinical conditions:
- Primary infection with VZV causes varicella (chickenpox), with subsequent lifetime latency of VZV in the dorsal root ganglion.9
- Reactivation of latent VZV typically results in a discrete rash in a dermatomal distribution, known as herpes zoster (shingles).7,8
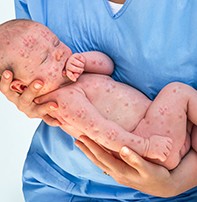
VZV can be transmitted via respiratory droplets before the development of the vesicular rash or via direct contact with vesicular fluid. The incubation period ranges from 10 to 21 days (most commonly 14 to 16 days). However, the incubation period may be prolonged up to 28 days in patients who receive zoster immunoglobulin.10 Individuals are infectious from one to two days before onset of the rash until all lesions have crusted over. Figure 2 shows a typical varicella rash with lesions at different stages of development.5,8
Complications
Chickenpox is typically a mild and self-limiting illness in healthy children. Complications occur in about 1% of cases and are more common in neonates, adults and individuals who are immunocompromised by another medical condition or its treatment.5,8
Complications of chickenpox include secondary bacterial infections (5%), pneumonia (1%), thrombocytopenia and neurological problems (1 to 3.5 in every 100,000 children aged under 14 years).11 Immunocompromised individuals are at higher risk of encephalomyelitis, cerebellar ataxia, hepatitis, arthritis, haemorrhagic nephritis, myocarditis and otitis media.7,11 Secondary bacterial infections, particularly invasive group A streptococcal or staphylococcal infections (e.g. cellulitis, myositis, necrotising fasciitis and toxic shock syndrome), are the most common cause of hospital admission in children.11,12
Management
The mainstay of treatment of chickenpox is supportive care, including hydration, antipyretics and pruritis management, as well as monitoring for complications.3,10 Calamine lotion and cool compresses can alleviate itching.10 Salicylates (aspirin) should be avoided because of the risk of Reye syndrome, an extremely rare acute encephalopathy with associated hepatic dysfunction. Reye syndrome is characterised by vomiting and confusion and may rapidly evolve to seizures, coma and death.12
Antiviral therapies licensed for chickenpox in children and adolescents include aciclovir and valaciclovir. Famciclovir is not used in children.13 Chickenpox in pregnant women, neonates, immunocompromised patients or patients with severe or complicated disease should prompt urgent referral for specialist and hospital care for consideration of antiviral therapy.14,15
Chickenpox is currently notifiable in all Australian states and territories apart from NSW.1
Prevention
The current recommended VZV vaccination schedule, adapted from the Australian Immunisation Handbook, is shown in the Table.14,16,17
Varicella vaccine
Varicella vaccine is a live attenuated vaccine that has been included in the Australian NIP since November 2005.7,8 It is given routinely as a single dose in combination with measles, mumps and rubella vaccines (MMRV) at 18 months of age.7,14 A second dose of varicella vaccine, not currently funded by the NIP, is recommended to improve protection and minimise the chance of breakthrough disease in children under 14 years of age.14
Single-dose varicella vaccine was shown to be 81% effective in preventing any chickenpox and 98% effective in preventing moderate to severe disease.18 Effectiveness in preventing any chickenpox rises to 92% after two doses of varicella vaccine. Varicella vaccine also reduces the risk of herpes zoster among vaccinated children.17-19 However, the rate of primary vaccine failure ranges from 9% to 14% after one dose, and the duration of protection after one dose has not been fully established.18
Varicella vaccination is also strongly recommended for: 7
- nonimmune adolescents
- adults at high risk of exposure to chickenpox, such as healthcare workers and childcare workers
- nonimmune women before pregnancy parents
- nonimmune household contacts of immunocompromised people (to minimise the risk of VZV transmission to the susceptible person).
In the absence of contraindications, varicella vaccine can be given for postexposure prophylaxis within five days, preferably three days, of exposure to varicella to nonimmune age-eligible children and adults.14
Varicella vaccine contains live attenuated virus. It is contraindicated for people who have had anaphylaxis to a dose or any component of the varicella-containing vaccine, pregnant women and people who are immunocompromised.7,14 A detailed list of medical conditions and therapies considered to cause severe immunosuppression can be found in the Australian Immunisation Handbook.14
Zoster vaccine
A single-dose live attenuated zoster vaccine is available in Australia and recommended for immunocompetent adults aged 60 years and over.17 It is currently funded under the National Shingles Vaccination Program for people aged 70 years, with catch up for those aged 71 to 79 years (until October 2021).17 Zoster vaccine contains 14 times the amount of live attenuated VZV compared with that in varicella vaccine.17 It is estimated to prevent 41% of cases of shingles and two-thirds of cases of postherpetic neuralgia in people aged 70 to 79 years.17
A prevaccination checklist, available on the Department of Health website, can help immunisation providers screen for contraindications to zoster vaccine. The checklist needs to be completed before administration of the vaccine (www.health.gov.au/news/clinical-update-pre-vaccination-checklist-for-zostavax-administration).20
Zoster immunoglobulin
Postexposure prophylaxis with zoster immunoglobulin (known as ZIG or VZIG) provides some short-term passive immunity to VZV. It is recommended for people who have substantially higher risk of complications from chickenpox, including:
- people who are severely immunocompromised
- susceptible pregnant women
- neonates, particular those who are preterm or whose mothers develop primary chickenpox within seven days before or up to 28 days after delivery.14,15
Zoster immunoglobulin is most effective if given within 96 hours of exposure, but some benefit may be conferred if given up to 10 days after exposure.12,14 Further information on specific indications for zoster immunoglobulin can be found in the Australian Immunisation Handbook.14
Contact precautions
People with chickenpox should be excluded from childcare, preschool, school and work until all vesicles have dried or for at least five days after the eruption first appears. They should also avoid contact with high-risk susceptible people, such as neonates, pregnant women and immunocompromised individuals.4 In healthcare settings, contact and airborne precautions should be implemented for patients with chickenpox.12
Conclusion
Despite a substantial reduction in morbidity and mortality from varicella following universal childhood varicella vaccination in Australia, this infection continues to cause serious (and sometimes fatal) complications in neonates, adults and people who are immunocompromised.21 GPs and other community health providers have an important role in identifying people who have chickenpox or have been exposed to chickenpox, especially high-risk individuals who require counselling, prophylaxis and potentially referral to hospital for further management. Practice points on varicella for GPs are summarised in the Box. MT