What’s the diagnosis?
A rapidly progressing, blistering eruption in a febrile patient
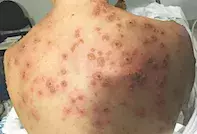
Case presentation
A 32-year-old primary school teacher presents to the accident and emergency department with fever associated with a painful and itchy blistering eruption. The lesions involve her trunk, arms, legs, face and scalp (Figure). The illness started with a generalised maculopapular eruption three days ago and progressed rapidly, with lesions cropping every few hours. She feels fatigued and mildly nauseated but is not significantly unwell.
Differential diagnoses
Differential diagnoses to consider in a patient with an evolving, generalised blistering eruption with fever include the following.
- Sweet’s syndrome (acute febrile neutrophilic dermatosis). Sweet’s syndrome is a rare neutrophilic dermatosis characterised by abrupt onset of a generalised, tender, erythematous eruption in an unwell, febrile patient. It can occur at any age, with peak onset between the fourth and seventh decades of life, and has a predominance in females.1 Triggers include occult malignancy, infection, drugs (e.g. NSAIDs, frusemide, trimethoprim-sulfamethoxazole), inflammatory bowel disease, autoimmune disorders (e.g. systemic lupus erythematosus) and pregnancy.1 Lesions in Sweet’s syndrome are typically nonpruritic, tender erythematous papules that may coalesce into plaques; these can exhibit pseudovesicular and targetoid morphology involving cutaneous surfaces of head, neck and upper limbs, sparing mucosal surfaces.1 Histopathology shows diffuse nodular perivascular neutrophilic infiltrate without evidence of vasculitis.1
- Pityriasis lichenoides et varioliformis acute (PLEVA). Patients with this condition are usually well and develop diffuse recurrent crops of pruritic and painful erythematous, purpuric papules that may form crusts, vesicles, pustules or ulcers. PLEVA has associations with infections (e.g. HIV, parvovirus B19) and drugs (e.g. 5-fluorouracil, radiocontrast iodide).2 Histopathology shows predominantly lymphocytic perivascular interface dermatitis, with an absence of vascular fibrinoid necrosis.2
- Staphylococcal folliculitis. A superficial infection of the hair follicle is most commonly due to Staphylococcus aureus, which results in pruritic and tender dome-shaped crusted papules and/or pustules on an erythematous base that may rupture to form a crust.3 These lesions commonly affect the face, scalp, back, chest, axillae and buttocks. Patients with localised folliculitis are usually well, with leukocytosis and neutrophilia. Bacterial swabs for microscopy, Gram stain and culture will confirm the diagnosis of staphylococcal folliculitis and dictate whether the causative organism is methicillin-sensitive or methicillin-resistant S. aureus.3 Involvement of deep soft tissue or large, coalescent erythematous papules in a febrile patient with recalcitrant staphylococcal folliculitis may indicate furunculosis or carbunculosis.3
- Bullous pemphigoid. Bullous pemphigoid results in a symmetrical distribution of subepidermal vesicles and bullae that tend to involve the flexural surfaces of extremities, lower trunk and mucosal surfaces of the oral cavity, nasopharynx, eyes and anogenital region.4 Bullous pemphigoid is most commonly seen in the eighth decade of life and can be triggered by trauma, drugs (e.g. spironolactone, NSAIDs, penicillins, ACE inhibitors), and is associated with neuropsychiatric disorders.4 The typical polymorphic clinical course of bullous pemphigoid occurs in two stages. An initial prodromal nonbullous period with nonspecific pruritus may be accompanied by erythematous papular and/or urticarial lesions. This is followed by a bullous phase characterised by the development of vesicles and bullae containing clear and/or blood-tinged fluid that can erupt causing erosions and crusts, with associated annular urticarial papules and plaques.4 Perilesional biopsy for direct immunofluorescence will show fine linear continuous deposits of IgG and C3 along the epidermal basement membrane, while blood tests may show a peripheral eosinophilia with IgG auto-antibodies to BP180 and BP230.4
- Erythema multiforme (EM). This condition is characterised by an abrupt onset of fixed pruritic and painful erythematous papules symmetrically affecting the upper extremities and face, which may evolve into the typical target lesions.5 The two clinical forms of EM (minor and major) are distinguished by the degree of mucosal involvement, which is more severe in EM major.5 Patients with EM minor are usually well, whereas systemic features such as fever, lethargy and arthralgias are almost always present in EM major.5 More than 90% of cases of EM are due to infections, most commonly HSV-1 and HSV-2; less common precipitants include drugs (e.g. NSAIDs, anticonvulsants) and systemic disease (e.g. inflammatory bowel disease, systemic lupus erythematosus).5 A history of herpes labialis is seen in half of patients with EM, which precedes the EM eruption from 3 to 14 days.
- Varicella infection. This is the correct diagnosis. Patients with varicella infection experience prodromal fever, myalgia and malaise, followed by an abrupt onset of a pruritic, erythematous, maculopapular eruption that initially appears on the scalp and face, with later involvement of the trunk, proximal extremities and mucosae, particularly the mucosa in the roof of the oral cavity.6 Lesions evolve into the characteristic small vesicles with a surrounding red halo within 12 hours, then form pustules with crusting; resolution occurs within 10 days of onset.6 In contrast to the benign and self-limiting disease course seen in children, varicella infection in adults is more severe, with increased cutaneous involvement and higher incidence of varicella pneumonia, hepatitis and encephalitis.6 Superimposed cutaneous bacterial infection, usually caused by Streptococcus pyogenes or S. aureus, is the most common complication.6 The diagnosis is primarily clinical, by clarifying exposure, previous varicella infection and/or vaccination, and is confirmed by PCR testing from a viral swab of an active vesicular lesion.6 Because of high levels of immunisation in the community, varicella is not seen as often as it once was.
Management and outcome
The patient was admitted to hospital because she was febrile and the medical team in the accident and emergency department were unsure of the diagnosis. A full blood count and biochemical testing were performed, and a raised white cell count and C-reactive protein were noted. Chest x-ray was performed to exclude pulmonary involvement.
The dermatology team was consulted the next day and made a diagnosis of varicella. The patient was started on oral valaciclovir and cephalexin to cover suspected bacterial superinfection. A bacterial swab subsequently revealed secondary infection with S. aureus, and cephalexin was therefore continued for 10 days. A viral swab confirmed the diagnosis of varicella.
The patient recovered completely, fortunately without the significant scarring that can be a complication of varicella. However, she suffered from a significant post-viral syndrome with severe fatigue and lethargy for six weeks and was unable to return to work for two months.
References
1. Cohen PR. Sweet's syndrome – a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis 2007; 2: 34.
2. Nair PS. A clinical and histopathological study of pityriasis lichenoides. Indian J Dermatol Venereol Leprol 2007; 73: 100-102.
3. Chiller K, Selkin BA, Murakawa GJ. Skin microflora and bacterial infections of the skin. J Investig Dermatol Symp Proc 2001; 6: 170-174.
4. Schmidt E, della Torre R, Borradori L. Clinical features and practical diagnosis of bullous pemphigoid. Immunol Allergy Clin North Am 2012; 32: 217-232.
5. Huff JC, Weston WL, Tonnesen MG. Erythema multiforme: a critical review of characteristics, diagnostic criteria, and causes. J Am Acad Dermatol 1983; 8: 763-775.
6. McCrary ML, Severson J, Tyring SK. Varicella zoster virus. J Am Acad Dermatol 1999; 41: 1-16.
Chickenpox

