Peer Reviewed
Gastroenterology clinic
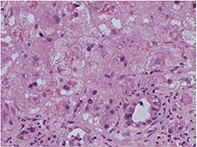
Amiodarone hepatoxicity
Abstract
Liver and lung toxicity associated with the use of amiodarone is uncommon but potentially life-threatening. Hepatotoxicity may be reversible if the drug is ceased as soon as toxicity is suspected.
Key Points
- Amiodarone hepatotoxicity can occur even on low-dose amiodarone therapy.
- There is a significant mortality and morbidity (approximately 1%).
- It can present as acute or chronic liver disease, early or late in the course of therapy (see the case studies in the box on page 58).
- The risk increases with higher doses and longer durations of therapy.
- The indication for amiodarone should be reviewed and other agents considered.
- Monitoring of liver function tests (one to three-monthly) is recommended throughout the duration of amiodarone therapy and for 12 months beyond cessation.
- Amiodarone should be ceased if the patient’s serum alanine aminotransferase (ALT) level is more than three times the normal for the testing pathology laboratory (i.e. is above about 150 U/L).
Remember
Get full access
Buy this article
Single article purchases are temporarily unavailable due to site maintenance.
If you would like to purchase an article during this time, please email us at [email protected] with the article details and we'll assist you directly. We'll also let you know when online purchasing is available again.
Thank you for your patience and understanding.
Already a subscriber? Login here.