Haemochromatosis – a clinician’s guide

Haemochromatosis is common in Australia and causes significant morbidity and mortality. Advances in the past few decades include improved understanding of its pathogenesis and a test for the most common genetic cause. Timely assessment through family screening and evaluation of individuals with suggestive biochemical or clinical features will enable early diagnosis and treatment.
- Haemochromatosis is a common condition that presents in general practice, especially in patients of European descent.
- Although several genetic mutations may cause haemochromatosis, homozygosity for the C282Y mutation in the HFE gene accounts for almost all clinically significant cases of hereditary haemochromatosis.
- The most common clinical manifestations of haemochromatosis are liver disease and arthritis; rates of dementia and malignancy, including hepatocellular carcinoma, cholangiocarcinoma and colorectal and breast cancer, are also increased.
- Identification of symptoms, family history and measurement of serum transferrin saturation and ferritin levels are the foundations of initial assessment for haemochromatosis.
- The cornerstone of management is regular phlebotomy to reduce the serum ferritin level to 50 to 100 mcg/L.
- Effective management is fundamental to normalising quality of life and survival, as well as regression of complications such as liver fibrosis.
Haemochromatosis describes a group of inherited disorders most often marked by reduced levels of the iron regulatory hormone hepcidin, which can result in iron overload. It is distinct from the secondary iron overload that occurs in conditions such as haemoglobinopathies (e.g. thalassaemia, sickle cell disease) and after repeated blood transfusions. Haemochromatosis is common in Australia. Significant advances over the past few decades include an understanding of the variable clinical expression of this condition and the crucial role of hepcidin, along with the ready availability of a genetic test for the principal cause, mutations in the HFE gene.
Nevertheless, haemochromatosis continues to be a significant cause of morbidity and premature death. Although the establishment of effective management can restore normal life expectancy, the nonspecific clinical and biochemical features of haemochromatosis are a challenge for diagnosis.
This article describes the most common form of haemochromatosis, HFE-related haemochromatosis (HH). It outlines a framework for GPs to identify and manage patients in their practice with this condition.
Genetic basis of HH
Although several genetic mutations can cause HH, at least 95% of cases are due to a homozygous mutation in the HFE gene, termed C282Y.1-4 This is a point substitution that results in the replacement of cysteine with tyrosine at position 282 of the resulting protein. This principal cause of HH is inherited in an autosomal recessive fashion. Other mutations in the HFE gene, including the point substitution H63D, are of little clinical consequence.
Epidemiology of HH
Haemochromatosis is marked by ethnic variability, with the highest rates of C282Y homozygosity affecting about one in every 150 to 220 individuals of European descent.2,4-7 Simple heterozygosity for C282Y or the more minor H63D variant in the HFE gene affects one in seven and one in three individuals, respectively, and does not cause any significant clinical disease. Likewise, heterozygosity for both the C282Y and H63D genotypes (‘compound heterozygosity’) represents an extremely low risk for clinical disease.2,4,8
According to the 2021 national census, 57% of the Australian population have European ancestry, suggesting that about 73,000 individuals in our community have HH.9
Pathogenesis of HH
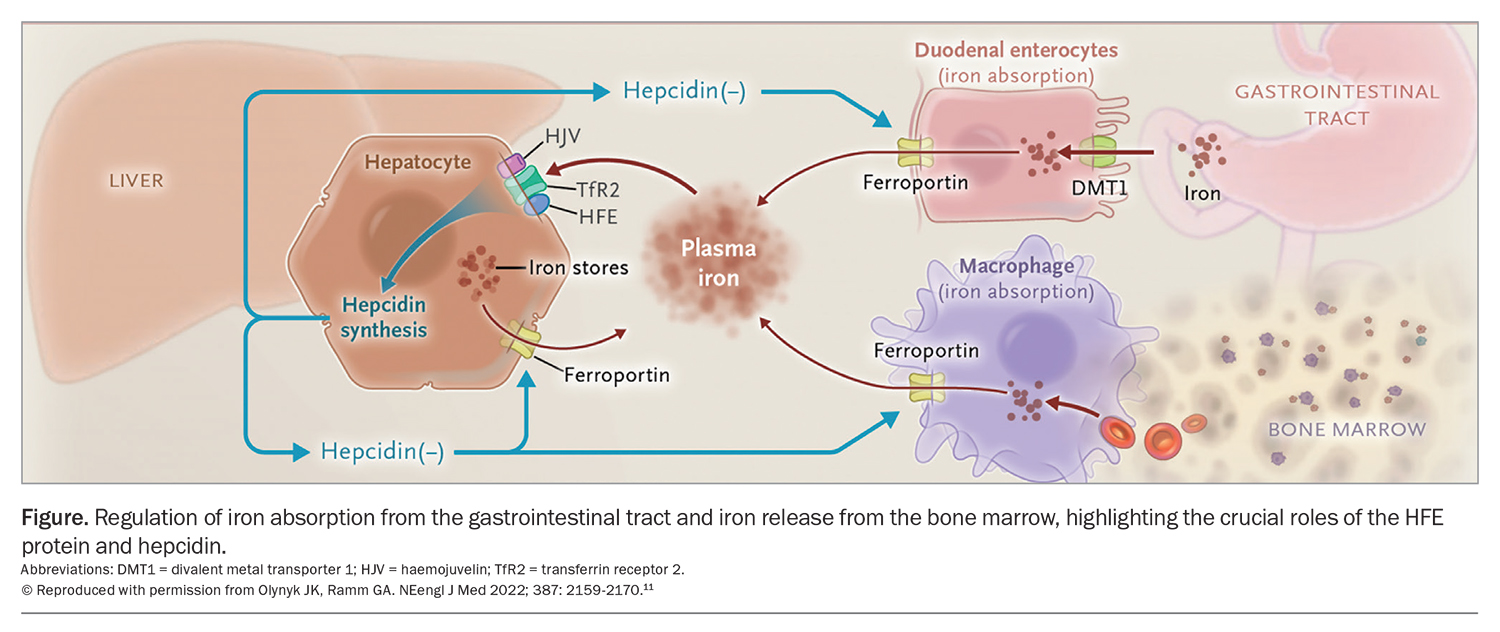
Iron is an important element for physiological function. Iron is absorbed by the enterocytes of the small intestine, predominantly in the duodenum and first section of the jejunum. It is most readily bioavailable from heme-containing sources (e.g. red meat). It is exported from enterocytes into the blood via the iron-export protein ferroportin. About 80 to 85% of absorbed iron is transported by transferrin for either storage in the liver or bone marrow or incorporation into haemoglobin or other proteins.10
The liver is an important organ in iron metabolism; it is not only the principal site of iron storage but also the key organ regulating iron absorption through the synthesis of the regulatory hormone, hepcidin (Figure).11 Increasing iron stores normally result in increased synthesis of hepcidin, which acts as a negative regulator, decreasing both iron absorption from the gastrointestinal tract and release of iron into the circulation from bone marrow stores through inhibition of ferroportin. Conversely, reduced iron stores result in lower hepcidin production, leading to increased iron absorption from the gastrointestinal tract and increased iron release from the bone marrow, as exemplified by the physiological response to iron-deficiency anaemia.
In patients with HH, the homozygous C282Y mutation abolishes the production of HFE protein. This protein is a key component of the regulatory pathway of hepcidin production, and its absence results in inappropriately low serum hepcidin levels. This leads to inappropriate increases in iron absorption from the intestine and iron release from the bone marrow, and substantially elevated total body iron stores in up to 40% of affected individuals.12 Other factors that can alter hepcidin expression and iron availability include infection, inflammation and hypoxia.13
Iron overload leads to oxidative stress-related tissue injury. This is responsible for the manifestations of HH at the organ level, including in the liver, heart and possibly joints.2,14
Clinical manifestations of HH
A major challenge in the diagnosis of HH relates to the variability of its biochemical and clinical manifestations. Further, these manifestations may not be progressive, which makes diagnosis and management challenging.4-7 The variable biochemical and clinical penetrance is most likely due to multiple genetic and environmental modifiers.15 Men are at higher risk than women, probably because of the protective effects of menstruation and pregnancy in reducing iron overload in women.16
Symptoms of HH are nonspecific and often have equal prevalence in individuals with and without HH.6,7 The most common symptom is fatigue, which is observed mainly in men with serum ferritin levels raised over 1000 mcg/L.6 The most frequent clinical morbidities are liver disease (advanced liver fibrosis or cirrhosis and primary liver cancer) and arthritis.3,8,17-19
Liver disease
Advanced liver fibrosis, cirrhosis and primary liver cancer (hepatocellular carcinoma [HCC] and cholangiocarcinoma) are the most significant liver disorders associated with HH.11 Advanced liver fibrosis or cirrhosis are rare under the age of 45 years in the absence of other liver comorbidities; they occur in about 8% of women and 25% of men with HH.1,3,4,17,20,21 Risk factors for advanced fibrosis include excess alcohol consumption, diabetes mellitus, arthritis, serum ferritin levels greater than 1000 mcg/L, platelet levels less than 200 × 109 cells/L, elevated aspartate aminotransferase (AST) levels, a liver iron concentration greater than 200 mcmol/g and total mobilisable iron stores by therapeutic phlebotomy of greater than 9.6 g.21-26 The risk of liver fibrosis also correlates with the duration of haemochromatosis and extent of total body iron levels. HH presenting with cirrhosis is uncommon; however, HH should always be considered in individuals of European descent with a new diagnosis of cirrhosis.
The risk of primary liver cancer is substantially increased in men, but not women, with HH; men with HH have a 12-fold increased lifetime risk of primary liver cancer of 7.2% compared with 0.6% in those without HFE mutations.27 The risk of primary liver cancer is greatest in people with advanced hepatic fibrosis or cirrhosis.18,19,28 These individuals should be recommended to undergo routine cancer surveillance with six-monthly liver ultrasound examination. Surveillance is generally continued lifelong as the risk of HCC remains while cirrhosis persists.2,3,8,17
Regression of advanced liver fibrosis has been associated with a significant reduction in the long-term risk of HCC, although cirrhosis persists after treatment in most cases.28 When regression is proven, clinicians may consider cessation of surveillance for HCC.
Arthritis
Arthritis is common in people with HH, affecting at least 24% of individuals, and is a major cause of disability and reduced quality of life.29,30 HH-associated arthritis presents similarly to degenerative osteoarthritis, with noninflammatory arthritis manifestations. Indeed, degenerative arthritis findings such as Heberden’s and Bouchard’s nodes with prominent involvement of the interphalangeal, knee and great toe metatarsophalangeal joints have similar frequency in people with HH as in people without the disorder.30 A helpful differentiating factor is the location of arthropathy; HH-associated arthritis affects the second to fifth metacarpophalangeal joints or bilateral large joints (radiocarpal, elbow, hip, knee and ankle joints) eight times more often than other types of arthritis.30
There is significant heterogeneity in the clinical manifestations of HH-associated arthritis; an unpredictable subset of patients are predisposed to arthritis, and it can occur at any point in the course of HH despite appropriate treatment.3,30 Risk factors for the development of arthritis include increasing age, advanced liver fibrosis, serum ferritin levels greater than 1000 mcg/L, and serum transferrin saturation greater than 50% for at least six years.31,32
Liver disease and arthritis tend to occur concomitantly, with arthritis more likely with a higher iron load or more advanced liver disease.33,34 A study found that 84% of participants with advanced liver fibrosis had arthritis, and 34% of those with HH and arthritis had advanced liver fibrosis, compared with 5% of those without arthritis. Thus, the absence of arthritis had a 95% negative predictive value for advanced liver fibrosis, making it a simple clinical tool for ascertaining the risk of significant liver disease.22
Other clinical manifestations of HH
Other conditions that have been reported predominantly in men with HH include dementia, osteoporosis, diabetes mellitus, hypogonadotrophic hypogonadism, pneumonia and cardiomyopathy.2,3,17,35,36 These conditions are usually managed as per the standard of care and in addition to the treatment of iron overload. There is also a twofold increased rate of colorectal cancer in both sexes and a twofold increased risk of breast cancer in women.35,37
Diagnostic approach
Screening for HH
General population screening for haemochromatosis has not been recommended because of variable and incomplete penetrance of the mutation and lack of proof of a resulting survival advantage.3,17,38,39 However, the recent report of significantly increased mortality in men who are homozygous for C282Y compared with those without HFE variants in the UK Biobank study supports re-examination of the utility of screening in susceptible male populations.27 Screening is indicated in first-degree relatives of probands, as discussed below.3,17
Recommended approach to clinical assessment
Common patient presentations to GPs requiring assessment for HH include:
- asymptomatic patients with a family history of HH
- asymptomatic patients with abnormal iron test results (elevated serum transferrin saturation, ferritin or aminotransferase levels)
- patients with symptoms of HH.2,3,17
Algorithms for the assessment and diagnosis of these three groups are outlined in Flowchart 1, Flowchart 2 and Flowchart 3. The general principles of assessment for HH are:
- assess for a genetic mutation likely to result in clinically significant iron overload
- stratify the risk of end-organ pathology, particularly advanced liver disease and arthritis.
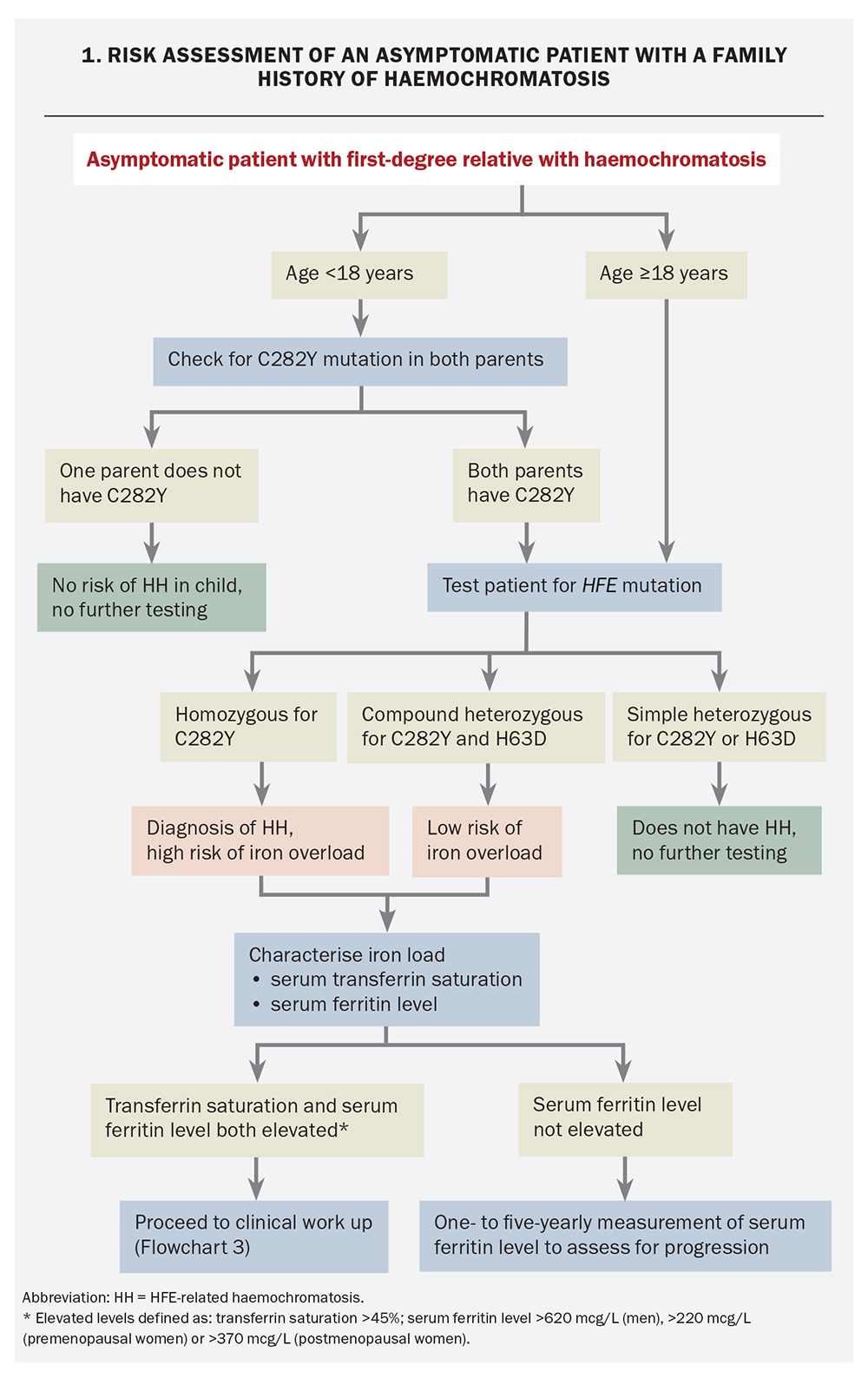
Asymptomatic patients with a family history of HH
For individuals under the age of 18 years with a family history of HH, the optimal approach is to determine the genotype of both parents as disease onset is extremely rare before 18 years (Flowchart 1).3,17,40 If one of the parents does not have the C282Y mutation then there is no risk of C282Y homozygosity in the offspring.3,40,41 If the H63D mutation is found in one parent (without C282Y), there is no strong recommendation to follow up the patient as compound heterozygosity for C282Y plus H63D carries an extremely low risk for development of iron overload.
Patients for whom genotyping of the other parent is not available and those who are over the age of 18 years should undergo HFE genetic testing at the earliest convenience. This is to assess for:
- C282Y homozygosity (high risk of iron overload)
- compound heterozygosity for C282Y and H63D (very low risk of iron overload)
- simple heterozygosity for C282Y or H63D (no increased risk of iron overload).
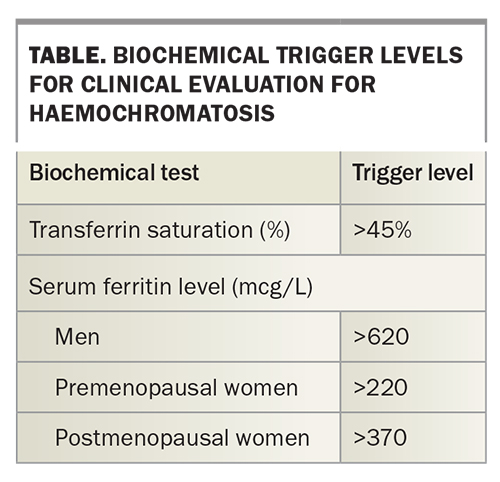
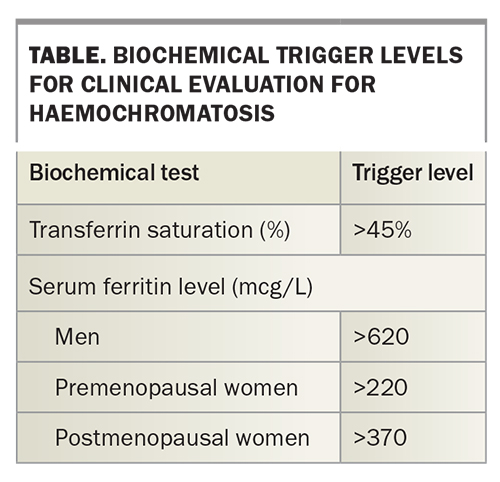
Serum transferrin saturation and ferritin levels should be measured in those who are homozygous for C282Y or compound heterozygous for C282Y and H63D. Patients with elevated transferrin saturation or serum ferritin levels (as defined in the Table) should undergo further work up as for those with HH symptoms (see below).42,43 If serum ferritin levels are not elevated then measurement of serum ferritin levels should be repeated after one to five years to assess for disease progression.3,17
In individuals who are homozygous for C282Y and aged 55 years or older and whose serum ferritin levels are within the reference range, surveillance can cease as it is highly unlikely that iron overload will ever evolve.44 In women, serum ferritin levels generally plateau 10 to 20 years after menopause at no greater than 400 mcg/L, a level too low to result in disease.16
Individuals who are homozygous for C282Y should be advised about family screening of first-degree relatives and standard-of-care colorectal and breast cancer surveillance.37,40
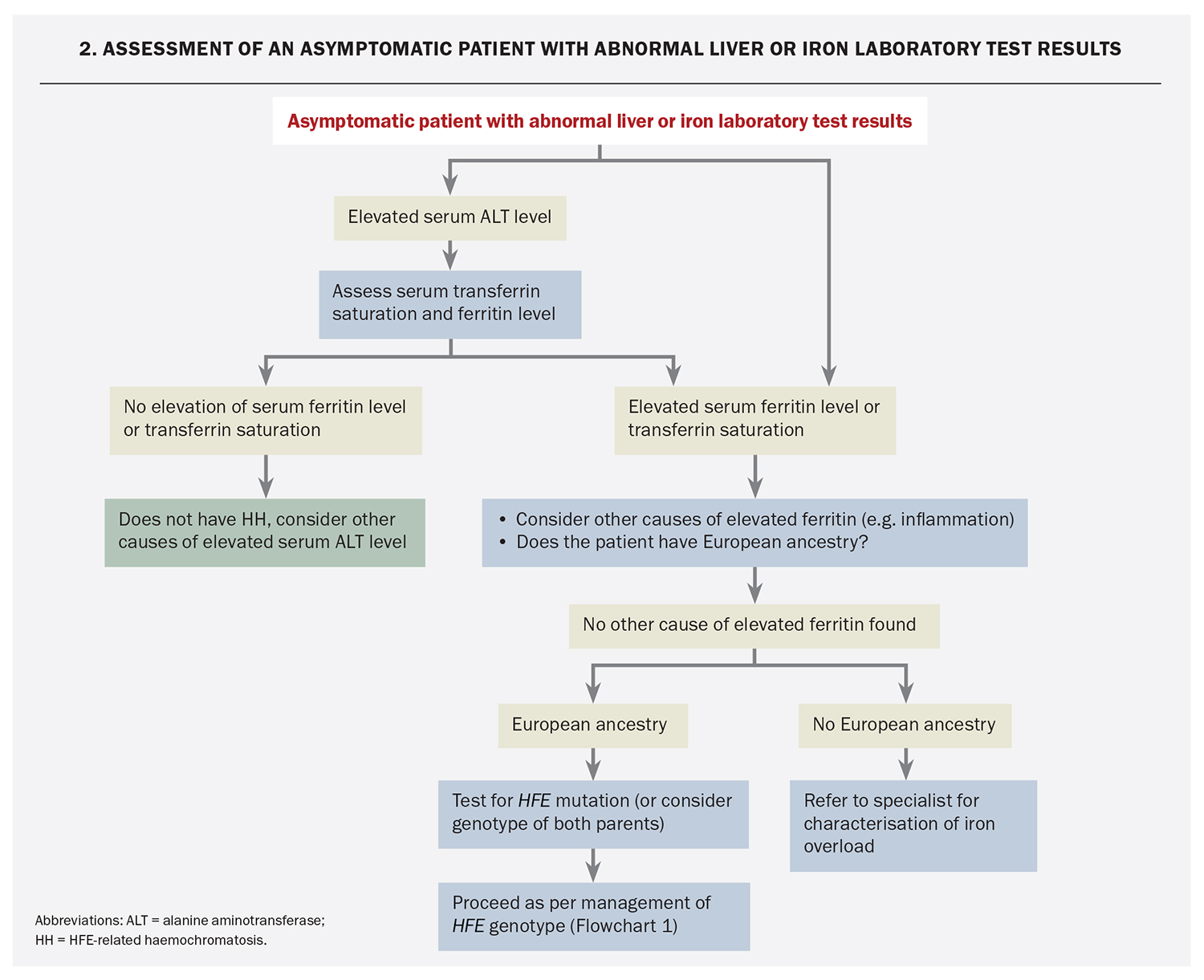
Asymptomatic patients with elevated serum transferrin saturation, ferritin or liver biochemistry results
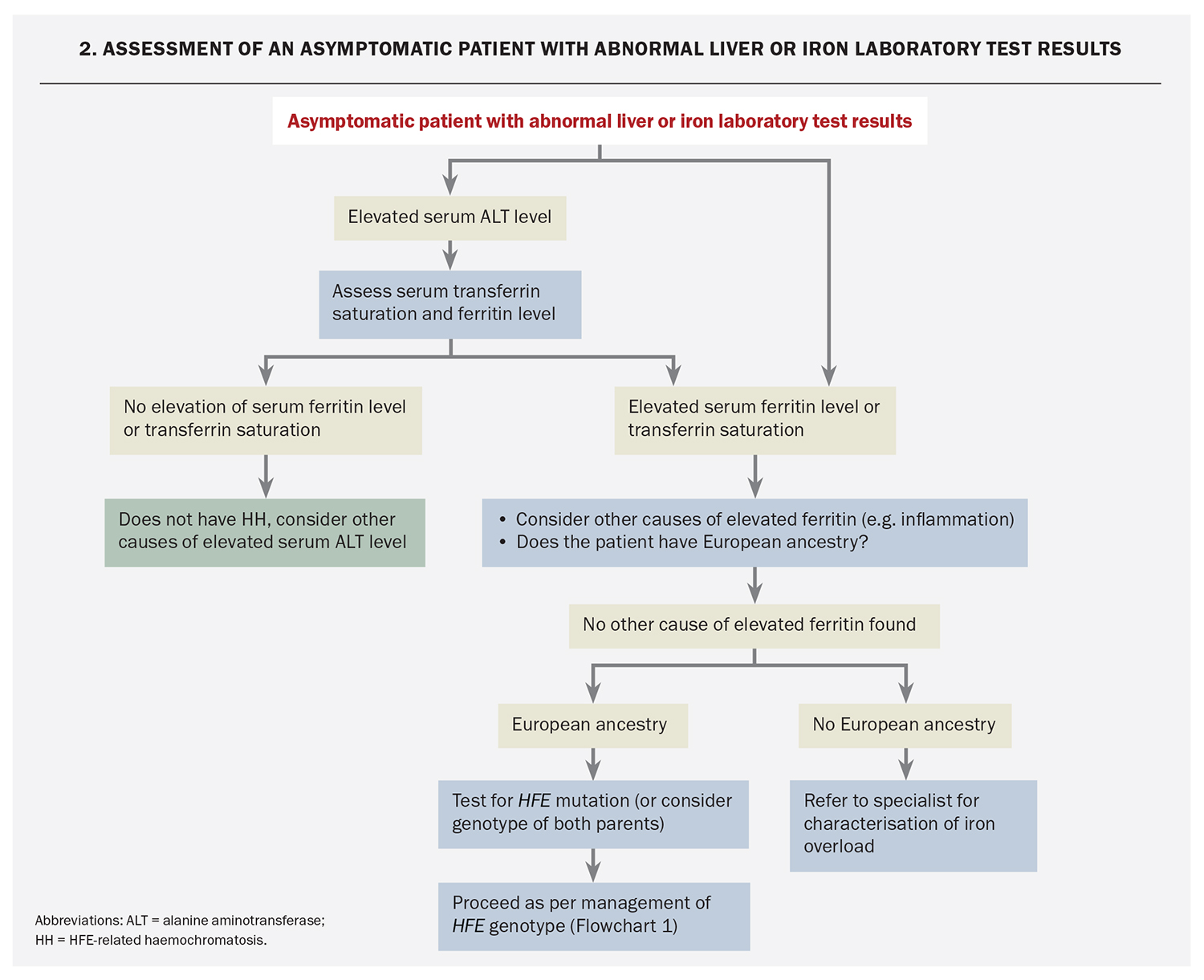
Elevated serum transferrin saturation or ferritin or aminotransferase levels are common in the general population. Elevated serum transferrin saturation or ferritin levels as defined in the Table are found in up to 6% of adults.4-7,42 An elevated serum alanine aminotransferase (ALT) level is seen in up to 14% of adults.45,46 Most people with this abnormality do not have HH. However, individuals with an abnormal ALT level should have serum transferrin saturation and ferritin levels measured (Flowchart 2).
If serum transferrin saturation and ferritin levels are elevated, the most appropriate investigation for those with European ancestry is testing for the HFE C282Y mutation. Those who are not of European ancestry and those found to be homozygous for C282Y should be evaluated as for patients with HH symptoms (see below).
Clinically significant iron overload is rare in people of European ancestry who are not homozygous for C282Y, even in the presence of elevated serum transferrin saturation or ferritin levels.47 An elevated serum ferritin level is thus not diagnostic on its own and requires confirmation with another method before iron overload is diagnosed, as described below (Quantification of Liver Iron).
For those whose serum transferrin saturation and ferritin level are not elevated, HH is unlikely. Alternative causes of ALT derangement should be sought.
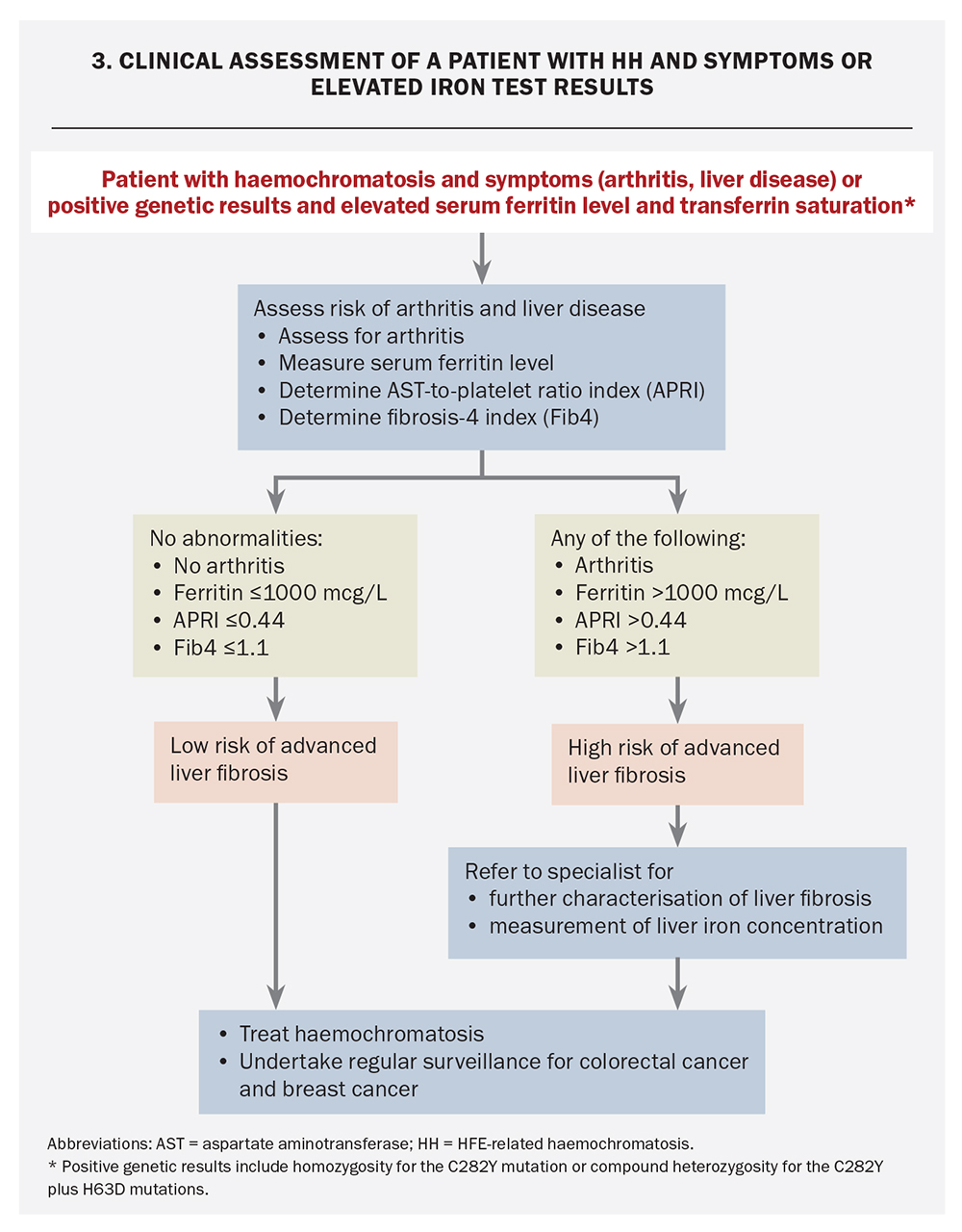
Patients with HH symptoms
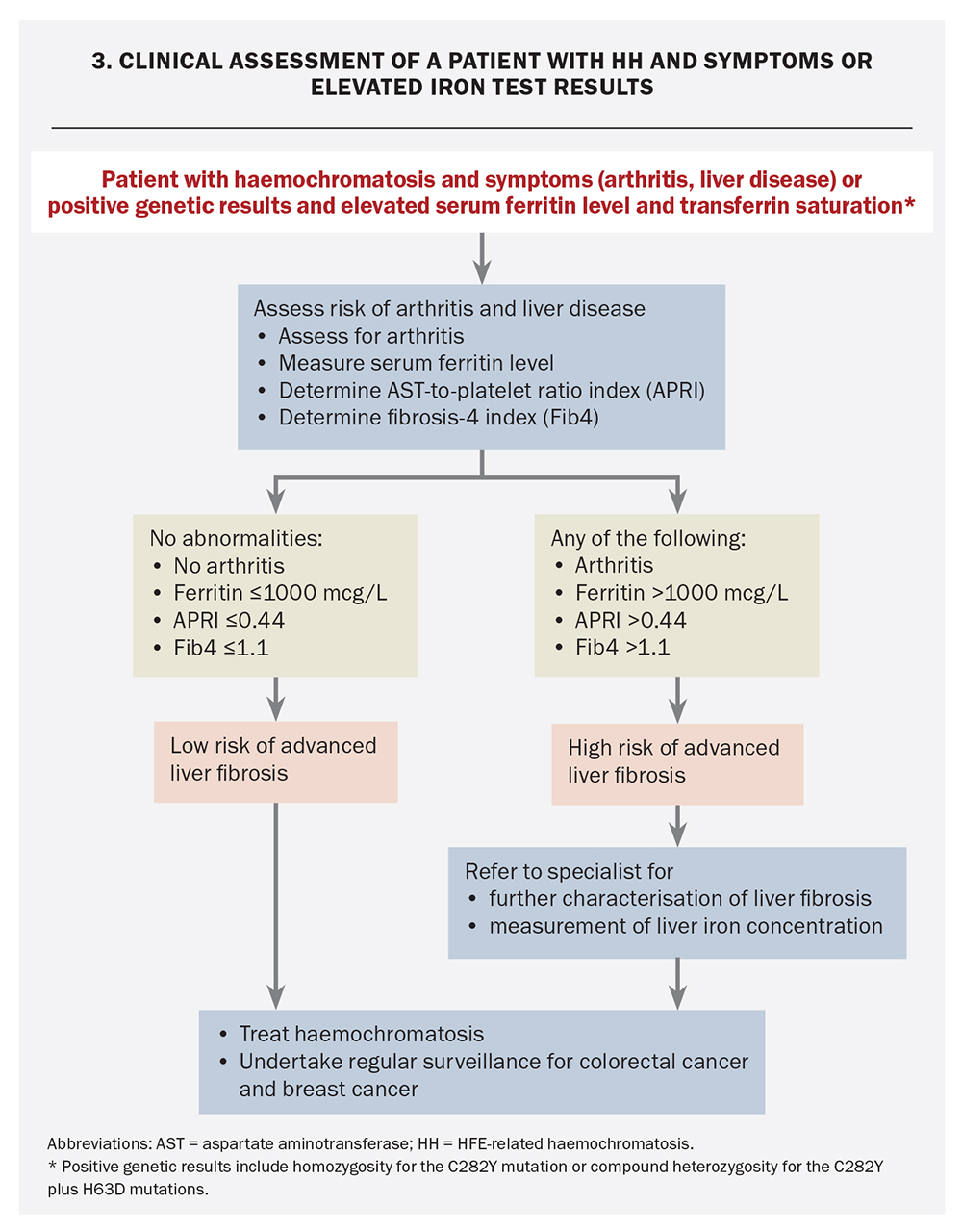
Patients who have symptoms consistent with HH should undergo clinical assessment to determine the risk of end-organ damage, especially liver disease and arthritis (Flowchart 3). The risks of arthritis, advanced liver fibrosis, cirrhosis and the subsequent development of primary liver cancer increase with progressive iron loading, particularly when the serum ferritin level exceeds 1000 mcg/L. HH arthritis is a useful clinical predictor for advanced liver fibrosis, as described above.22,33,34 The presence of additional risk cofactors for liver disease in people who are homozygous for C282Y may warrant assessment of liver disease at lower ferritin levels.3,17
Key components of the assessment of severity of liver disease are quantification of liver iron concentration and assessment of the liver fibrosis stage. These assessments should be routine in all patients with arthritis or serum ferritin levels greater than 1000 mcg/L and in those at risk because of additional cofactors, including unexplained hepatomegaly or elevated serum aminotransferase levels.
Quantification of liver iron
Increasing liver iron concentration is associated with an increased risk of advanced liver fibrosis and the development of cirrhosis. There are three ways to quantify liver iron concentration:
- invasively via liver biopsy, which is now rarely performed as more reliable methods of noninvasive measurement are available3,8,17,23,48
- retrospective calculation of iron removed based on the number and volume of therapeutic phlebotomies undertaken to reduce the serum ferritin level to 50 to 100 mcg/L17
- MRI quantification of the liver iron concentration.
Allowing for substantial heterogeneity in iron deposition within the liver and the attendant limitations of comparing biopsy with imaging methods, MRI provides good clinical utility for quantifying iron overload and thus is the preferred method.3,49-52 MRI is also accurate for quantifying myocardial iron deposition.53 In Australia, an automated system for MRI quantification of liver iron concentration (FerriSmart) has been approved by the TGA.51
Assessment of liver fibrosis
Assessment of the stage of liver fibrosis is crucial to management and prognostication for patients with HH. Further, in patients with cirrhosis, the finding of regression of advanced fibrosis with phlebotomy therapy can guide the requirement for ongoing HCC surveillance.24
Methods to assess liver fibrosis include:
- liver biopsy - although previously considered a gold standard, biopsy is now used less often because of its invasive nature and the development of validated noninvasive serum biomarker panels
- noninvasive serum biomarker panels
– AST-to-platelet ratio index (APRI = [AST/AST upper limit of normal] × [100/platelet concentration])
– fibrosis-4 index (Fib4 = [age x AST]/[platelets × √ALT]) - transient elastography.54,55
Detection of advanced liver fibrosis and cirrhosis is crucial for defining prognosis and the risk of HCC. The noninvasive scores APRI and Fib4 accurately detect liver biopsy-diagnosed advanced liver fibrosis in patients with HH with an accuracy of over 80%, with APRI also useful for monitoring fibrosis regression during treatment.54 The Hepascore (an algorithm based on age, sex and serum levels of bilirubin, alpha2-macroglobulin, hyaluronic acid, gamma-glutamyl transferase) and transient elastography were shown in a recent study to be more limited in their clinical utility as they may underdiagnose advanced liver fibrosis.55
Individuals with noninvasive marker values consistent with advanced liver fibrosis may be recommended to undergo liver biopsy for definitive diagnosis when clinically appropriate.3,17 All individuals with advanced liver fibrosis or cirrhosis are at high risk of primary liver cancer and require cancer surveillance.3,8,17,18,28
Management
The aim of management in patients with HH is to return total body iron stores to the low-normal range and to minimise or reverse clinical complications of HH. All patients with HH and elevated serum ferritin levels should be treated, as this has been shown to result in clinical improvement.3,17,56 Disease manifestations of HH should also be managed as per the standard of care for the particular problem.
Dietary treatment
Individuals with HH should adopt a healthy lifestyle, including maintaining normal body weight and limiting alcohol consumption.57 Dietary iron intake does not need to be restricted, as this has little or no proven benefit in routine management.3,8,17,58 However, iron supplements should be avoided.
Phlebotomy
Phlebotomy is the mainstay of treatment for patients with HH. Phlebotomy is recommended in all those who are homozygous for C282Y and have elevated serum ferritin levels. There is no good evidence supporting phlebotomy treatment for individuals with unexplained elevated ferritin levels caused by other conditions, such as fatty liver disease, or where iron overload has not been definitively proven by confirmatory testing as described above (Quantification of Liver Iron).59,60
Patients with HH and serum ferritin levels greater than 1000 mcg/L have a markedly increased risk of advanced liver fibrosis or cirrhosis. Treatment with phlebotomoy improves fatigue, cognition and liver fibrosis.24,28,54,56,61 Improvements in liver fibrosis occur across the spectrum of fibrosis.24,28,54,61 Cirrhosis regresses with adequate phlebotomy therapy in up to 23% of individuals; 18% regressing to levels below the threshold associated with the long-term risk of primary liver cancer.28
Although phlebotomy is an effective and safe therapy, some HH individuals find it difficult to comply for reasons such as lack of motivation, needle phobia and difficult venous access.62 Further, some patients have symptoms caused by hypovolaemia after phlebotomy or problems such as cardiac morbidity, hypoproteinaemia and thrombocytopenia.63 In these situations, alternative approaches including erythrocytapheresis or chelation therapy may be considered.
Erythrocytapheresis is a method of red blood cell removal where whole blood is drawn from the patient, centrifuged to separate it into plasma and red cells, and the plasma is returned to the patient. Compared with phlebotomy, erythrocytapheresis can reduce the time to achieve acceptable serum ferritin levels and reduce symptoms of hypovolaemia. It requires specialised equipment and is, therefore, more costly and less accessible than phlebotomy.64-66 It should be considered by specialists in patients with contraindications or difficulties with phlebotomy.
Chelation therapy, most commonly oral deferasirox, may be considered for patients with HH who cannot tolerate other iron removal therapies.67
Phlebotomy treatment has two distinct phases:
- induction, which has a target serum ferritin level of 50 to 100 mcg/L
- maintenance, which aims to maintain serum ferritin levels at 50 to 100 mcg/L.
An electronic referral tool for phlebotomy, confirming eligibility and prescribing the frequency of therapy is the standard of care in Australia and available at the website of the Australian Red Cross Lifeblood service (https://highferritin.transfusion.com.au/). Blood products prepared from individuals with HH can be safely used for transfusion purposes.
An unexplained reduction in the need for phlebotomy should be investigated as it may be due to occult blood loss. On occasion, no explanation for the reduced need can be identified suggesting there may be variation in phenotypic expression of the genetic mutation within individuals.68
Induction phase
Treatment in the induction phase usually involves weekly phlebotomy until an endpoint serum ferritin level of 50 to 100 mcg/L is reached.3,17 Less frequent phlebotomy therapy may be necessary in some patients who cannot tolerate weekly treatments. Measurement of haemoglobin is recommended at each phlebotomy, whereas serum ferritin level can be measured at every fourth phlebotomy during the early induction phase, increasing to every phlebotomy when the serum ferritin level reaches 200 mcg/L.
Maintenance phase
After the target serum ferritin level has been achieved, the maintenance phase commences to stabilise the level. This most often involves three-monthly phlebotomy, but the required frequency is highly variable and needs to be individualised and titrated to target serum ferritin levels. Serum ferritin levels may need to be monitored only once or twice yearly after the phlebotomy requirements are stabilised. Noncompliance with venesection is more common in the maintenance phase, and noncompliant patients are at risk of reaccumulating iron and associated complications.62
Prognosis
Recent population studies have shown that men, but not women, homozygous for C282Y have a significantly increased mean risk of death by the age of 75 years of 19.5% compared with 15.1% for the control group.27 The development of chronic liver disease further reduces long-term survival. Early phlebotomy treatment before the onset of cirrhosis results in normal survival compared with subjects matched for age and sex. Given the potential for reversal of advanced hepatic fibrosis and cirrhosis with phlebotomy, survival in these individuals is also likely to be substantially improved by treatment.
Conclusion
HH is a common disorder with variable clinical expression in men and women, which if left untreated can lead to significantly increased morbidity and mortality. Timely assessment through family screening and evaluation of individuals with suggestive biochemical or clinical features will detect disease at the earliest opportunity. Men have greater mortality and morbidity than women; the main clinical issues are liver disease, arthritis and dementia. All individuals with HH and elevated serum ferritin levels should receive treatment. All adults with HH should be informed of their increased risk of breast and colorectal cancer and receive appropriate screening. Long-term follow-up is best managed by community-based GPs in partnership with the patient, and in conjunction with supporting specialists to ensure optimal patient outcomes. Individuals with elevated serum ferritin levels who do not have HH or another proven iron-overload disorder do not need monitoring of serum ferritin levels or phlebotomy treatment. MT
COMPETING INTERESTS: Professor Olynyk is Chief Medical Officer for Resonance Health, a TGA-approved provider of MRI measurement of liver iron levels (FerriSmart) and received travel support from Resonance Health to attend a conference. He provides GP education talks for AstraZeneca, Pfizer and Pharmacosmos and medical advice to AstraZeneca, Australian Red Cross Lifeblood and Haemochromatosis Australia. Dr Chen: None.
References
1. Bacon BR, Olynyk JK, Brunt EM, Britton RS, Wolff RK. HFE genotype in patients with hemochromatosis and other liver diseases. Ann Intern Med 1999; 130: 953-962.
2. Brissot P, Pietrangelo A, Adams PC, de Graaff B, McLaren CE, Loreal O. Haemochromatosis. Nat Rev Dis Primers 2018; 4: 18016.
3. Kowdley KV, Brown KE, Ahn J, Sundaram V. ACG clinical guideline: hereditary hemochromatosis. Am J Gastroenterol 2019; 114: 1202-1218.
4. Olynyk JK, Cullen DJ, Aquilia S, Rossi E, Summerville L, Powell LW. A population-based study of the clinical expression of the hemochromatosis gene. N Engl J Med 1999; 341: 718-724.
5. Adams PC, Reboussin DM, Barton JC, et al. Hemochromatosis and iron-overload screening in a racially diverse population. N Engl J Med 2005; 352: 1769-1778.
6. Allen KJ, Gurrin LC, Constantine CC, et al. Iron-overload-related disease in HFE hereditary hemochromatosis. N Engl J Med 2008; 358: 221-230.
7. Beutler E, Felitti VJ, Koziol JA, Ho NJ, Gelbart T. Penetrance of 845G--> A (C282Y) HFE hereditary haemochromatosis mutation in the USA. Lancet 2002; 359: 211-218.
8. Girelli D, Busti F, Brissot P, Cabantchik I, Muckenthaler MU, Porto G. Hemochromatosis classification: update and recommendations by the BIOIRON Society. Blood 2022; 139: 3018-3029.
9. Australian Bureau of Statistics. Population: census. Canberra: ABS; 2022. Available online at: https://www.abs.gov.au/statistics/people/population (accessed March 2023).
10. Bacon BR, Tavill AS. Role of the liver in normal iron metabolism. Semin Liver Dis 1984; 4: 181-192.
11. Olynyk JK, Ramm GA. Hemochromatosis. N Engl J Med 2022; 387: 2159-2170.
12. Bridle KR, Frazer DM, Wilkins SJ, et al. Disrupted hepcidin regulation in HFE-associated haemochromatosis and the liver as a regulator of body iron homoeostasis. Lancet 2003; 361: 669-673.
13. Rishi G, Wallace DF, Subramaniam VN. Hepcidin: regulation of the master iron regulator. Biosci Rep 2015; 35: e00192.
14. Ramm GA, Ruddell RG. Iron homeostasis, hepatocellular injury, and fibrogenesis in hemochromatosis: the role of inflammation in a noninflammatory liver disease. Semin Liver Dis 2010; 30: 271-287.
15. Anderson GJ, Bardou-Jacquet E. Revisiting hemochromatosis: genetic vs. phenotypic manifestations. Ann Transl Med 2021; 9: 731.
16. Warne CD, Zaloumis SG, Bertalli NA, et al. HFE p.C282Y homozygosity predisposes to rapid serum ferritin rise after menopause: a genotype-stratified cohort study of hemochromatosis in Australian women. J Gastroenterol Hepatol 2017; 32: 797-802.
17. Bacon BR, Adams PC, Kowdley KV, Powell LW, Tavill AS, American Association for the Study of Liver Diseases. Diagnosis and management of hemochromatosis: 2011 practice guideline by the American Association for the Study of Liver Diseases. Hepatology 2011; 54: 328-343.
18. Niederau C, Fischer R, Purschel A, Stremmel W, Haussinger D, Strohmeyer G. Long-term survival in patients with hereditary hemochromatosis. Gastroenterology 1996; 110: 1107-1119.
19. Niederau C, Fischer R, Sonnenberg A, Stremmel W, Trampisch HJ, Strohmeyer G. Survival and causes of death in cirrhotic and in noncirrhotic patients with primary hemochromatosis. N Engl J Med 1985; 313: 1256-1262.
20. Scheuer PJ. Classification of chronic viral hepatitis: a need for reassessment. J Hepatol 1991; 13: 372-374.
21. Chin J, Powell LW, Ramm LE, Ayonrinde OT, Ramm GA, Olynyk JK. Utility of hepatic or total body iron burden in the assessment of advanced hepatic fibrosis in HFE hemochromatosis. Sci Rep 2019; 9: 20234.
22. Andersson L, Powell LW, Ramm LE, Ramm GA, Olynyk JK. Arthritis prediction of advanced hepatic fibrosis in HFE hemochromatosis. Mayo Clin Proc 2022; 97: 1649-1655.
23. Beaton M, Guyader D, Deugnier Y, Moirand R, Chakrabarti S, Adams P. Noninvasive prediction of cirrhosis in C282Y-linked hemochromatosis. Hepatology 2002; 36: 673-678.
24. Powell LW, Dixon JL, Ramm GA, et al. Screening for hemochromatosis in asymptomatic subjects with or without a family history. Arch Intern Med 2006; 166: 294-301.
25. Wood MJ, Crawford DHG, Wockner LF, Powell LW, Ramm GA. Serum ferritin concentration predicts hepatic fibrosis better than hepatic iron concentration in human HFE-haemochromatosis. Liver Int 2017; 37: 1382-1388.
26. Wood MJ, Powell LW, Dixon JL, Ramm GA. Clinical cofactors and hepatic fibrosis in hereditary hemochromatosis: the role of diabetes mellitus. Hepatology 2012; 56: 904-911.
27. Atkins JL, Pilling LC, Masoli JAH, et al. Association of hemochromatosis HFE p.C282Y homozygosity with hepatic malignancy. JAMA 2020; 324: 2048-2057.
28. Bardou-Jacquet E, Morandeau E, Anderson GJ, et al. Regression of fibrosis stage with treatment reduces long-term risk of liver cancer in patients with hemochromatosis caused by mutation in HFE. Clin Gastroenterol Hepatol 2020; 18: 1851-1857.
29. Adams PC, Speechley M. The effect of arthritis on the quality of life in hereditary hemochromatosis. J Rheumatol 1996; 23: 707-710.
30. Carroll GJ, Breidahl WH, Bulsara MK, Olynyk JK. Hereditary hemochromatosis is characterized by a clinically definable arthropathy that correlates with iron load. Arthritis Rheum 2011; 63: 286-294.
31. Bardou-Jacquet E, Laine F, Guggenbuhl P, et al. Worse outcomes of patients with HFE hemochromatosis with persistent increases in transferrin saturation during maintenance therapy. Clin Gastroenterol Hepatol 2017; 15: 1620-1627.
32. Carroll GJ, Breidahl WH, Olynyk JK. Characteristics of the arthropathy described in hereditary hemochromatosis. Arthritis Care Res (Hoboken) 2012; 64: 9-14.
33. Altes A, Ruiz A, Martinez C, et al. The relationship between iron overload and clinical characteristics in a Spanish cohort of 100 C282Y homozygous hemochromatosis patients. Ann Hematol 2007; 86: 831-835.
34. Valenti L, Fracanzani AL, Rossi V, et al. The hand arthropathy of hereditary hemochromatosis is strongly associated with iron overload. J Rheumatol 2008; 35: 153-158.
35. Pilling LC, Tamosauskaite J, Jones G, et al. Common conditions associated with hereditary haemochromatosis genetic variants: cohort study in UK Biobank. BMJ 2019; 364: k5222.
36. Atkins JL, Pilling LC, Heales CJ, et al. Hemochromatosis mutations, brain iron imaging, and dementia in the UK Biobank Cohort. J Alzheimers Dis 2021; 79: 1203-1211.
37. Osborne NJ, Gurrin LC, Allen KJ, et al. HFE C282Y homozygotes are at increased risk of breast and colorectal cancer. Hepatology 2010; 51: 1311-1318.
38. Jacobs EM, Hendriks JC, Marx JJ, et al. Morbidity and mortality in first-degree relatives of C282Y homozygous probands with clinically detected haemochromatosis compared with the general population: the Hemochromatosis FAmily Study (HEFAS). Neth J Med 2007; 65: 425-433.
39. Whitlock EP, Garlitz BA, Harris EL, Beil TL, Smith PR. Screening for hereditary hemochromatosis: a systematic review for the U.S. Preventive Services Task Force. Ann Intern Med 2006; 145: 209-223.
40. Delatycki MB, Powell LW, Allen KJ. Hereditary hemochromatosis genetic testing of at-risk children: what is the appropriate age? Genet Test 2004; 8: 98-103.
41. Adams PC. Implications of genotyping of spouses to limit investigation of children in genetic hemochromatosis. Clin Genet 1998; 53: 176-178.
42. McKinnon EJ, Rossi E, Beilby JP, Trinder D, Olynyk JK. Factors that affect serum levels of ferritin in Australian adults and implications for follow-up. Clin Gastroenterol Hepatol 2014; 12: 101-108.e4.
43. Royal College of Pathologists of Australasia. Ferritin. Last updated 2021. Available online at: https://www.rcpa.edu.au/Manuals/RCPA-Manual/Pathology-Tests/F/Ferritin (accessed March 2023).
44. Gurrin LC, Osborne NJ, Constantine CC, et al. The natural history of serum iron indices for HFE C282Y homozygosity associated with hereditary hemochromatosis. Gastroenterology 2008; 135: 1945-1952.
45. Kim HC, Nam CM, Jee SH, Han KH, Oh DK, Suh I. Normal serum aminotransferase concentration and risk of mortality from liver diseases: prospective cohort study. BMJ 2004; 328: 983.
46. Ruhl CE, Everhart JE. Elevated serum alanine aminotransferase and gamma-glutamyltransferase and mortality in the United States population. Gastroenterology 2009; 136: 477-485.e11.
47. Olynyk JK, Gan E, Tan T. Predicting iron overload in hyperferritinemia. Clin Gastroenterol Hepatol 2009; 7: 359-362.
48. Olynyk JK, St Pierre TG, Britton RS, Brunt EM, Bacon BR. Duration of hepatic iron exposure increases the risk of significant fibrosis in hereditary hemochromatosis: a new role for magnetic resonance imaging. Am J Gastroenterol 2005; 100: 837-841.
49. Emond MJ, Bronner MP, Carlson TH, Lin M, Labbe RF, Kowdley KV. Quantitative study of the variability of hepatic iron concentrations. Clin Chem 1999; 45: 340-346.
50. Sarigianni M, Liakos A, Vlachaki E, et al. Accuracy of magnetic resonance imaging in diagnosis of liver iron overload: a systematic review and meta-analysis. Clin Gastroenterol Hepatol 2015; 13: 55-63.e5.
51. St Pierre TG, Clark PR, Chua-anusorn W, et al. Noninvasive measurement and imaging of liver iron concentrations using proton magnetic resonance. Blood 2005; 105: 855-861.
52. Gandon Y, Olivie D, Guyader D, et al. Non-invasive assessment of hepatic iron stores by MRI. Lancet 2004; 363: 357-362.
53. Anderson LJ, Holden S, Davis B, et al. Cardiovascular T2-star (T2*) magnetic resonance for the early diagnosis of myocardial iron overload. Eur Heart J 2001; 22: 2171-2179.
54. Chin J, Powell LW, Ramm LE, Hartel GF, Olynyk JK, Ramm GA. Utility of serum biomarker indices for staging of hepatic fibrosis before and after venesection in patients with hemochromatosis caused by variants in HFE. Clin Gastroenterol Hepatol 2021; 19: 1459-1468.e5.
55. Ong SY, Khoo T, Nicoll AJ, et al. Utility and limitations of Hepascore and transient elastography to detect advanced hepatic fibrosis in HFE hemochromatosis. Sci Rep 2021; 11: 14654.
56. Ong SY, Gurrin LC, Dolling L, et al. Reduction of body iron in HFE-related haemochromatosis and moderate iron overload (Mi-Iron): a multicentre, participant-blinded, randomised controlled trial. Lancet Haematol 2017; 4: e607-e614.
57. Powell LW, Seckington RC, Deugnier Y. Haemochromatosis. Lancet 2016; 388: 706-716.
58. Moretti D, van Doorn GM, Swinkels DW, Melse-Boonstra A. Relevance of dietary iron intake and bioavailability in the management of HFE hemochromatosis: a systematic review. Am J Clin Nutr 2013; 98: 468-479.
59. Valenti L, Fracanzani AL, Dongiovanni P, et al. A randomized trial of iron depletion in patients with nonalcoholic fatty liver disease and hyperferritinemia. World J Gastroenterol 2014; 20: 3002-3010.
60. Adams LA, Crawford DH, Stuart K, et al. The impact of phlebotomy in nonalcoholic fatty liver disease: a prospective, randomized, controlled trial. Hepatology 2015; 61: 1555-1564.
61. Prabhu A, Cargill T, Roberts N, Ryan JD. Systematic review of the clinical outcomes of iron reduction in hereditary hemochromatosis. Hepatology 2020; 72: 1469-1482.
62. Hicken BL, Tucker DC, Barton JC. Patient compliance with phlebotomy therapy for iron overload associated with hemochromatosis. Am J Gastroenterol 2003; 98: 2072-2077.
63. Rombout-Sestrienkova E, van Kraaij MG, Koek GH. How we manage patients with hereditary haemochromatosis. Br J Haematol 2016; 175: 759-770.
64. Rombout-Sestrienkova E, Brandts L, Koek GH, van Deursen C. Patients with hereditary hemochromatosis reach safe range of transferrin saturation sooner with erythrocytaphereses than with phlebotomies. J Clin Apher 2022; 37: 100-105.
65. Rombout-Sestrienkova E, Koek GH, Neslo R, et al. Course of iron parameters in HFE-hemochromatosis patients during initial treatment with erythrocytapheresis compared to phlebotomy. J Clin Apher 2016; 31: 564-570.
66. Rombout-Sestrienkova E, Winkens B, Essers BA, et al. Erythrocytapheresis versus phlebotomy in the maintenance treatment of HFE hemochromatosis patients: results from a randomized crossover trial. Transfusion 2016; 56: 261-270.
67. Cancado R, Melo MR, de Moraes Bastos R, et al. Deferasirox in patients with iron overload secondary to hereditary hemochromatosis: results of a 1-yr Phase 2 study. Eur J Haematol 2015; 95: 545-550.
68. Adams PC. Factors affecting the rate of iron mobilization during venesection therapy for genetic hemochromatosis. Am J Hematol 1998; 58: 16-19.