Contraception in 2021: an update
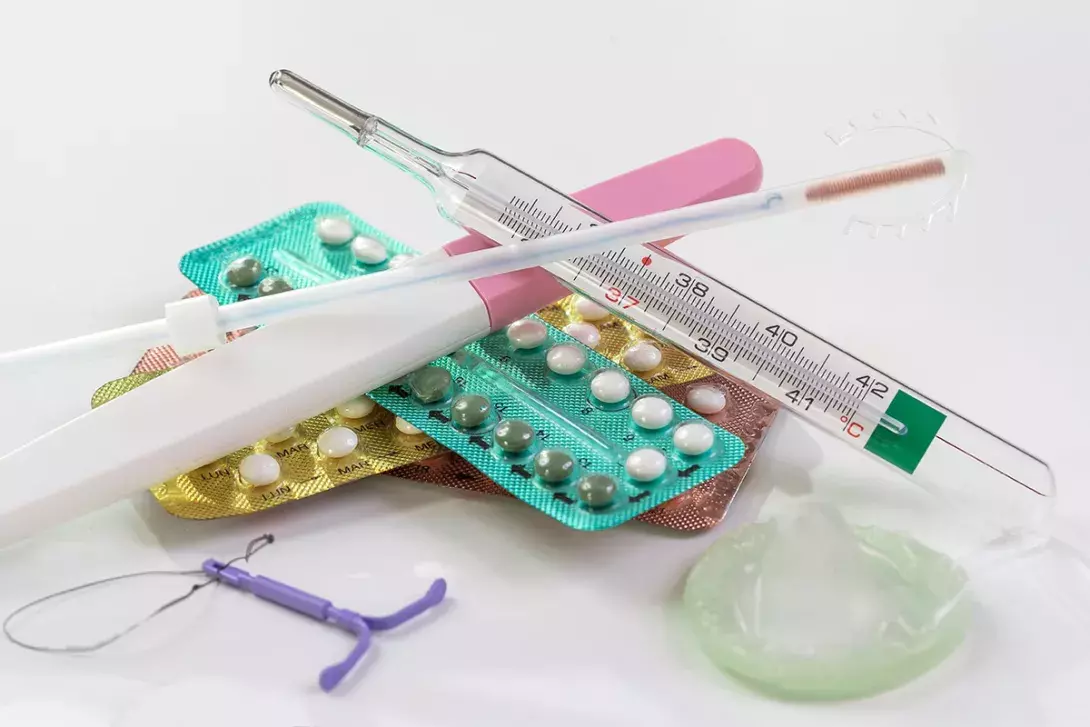
Several long- and shorter-acting contraceptive methods are available in Australia, including traditional hormonal and nonhormonal intrauterine devices (IUDs) and combined hormonal oral contraceptives. The low-dose hormonal IUD and a progestogen-only pill that reliably inhibits ovulation are newer options. GPs should be aware of the advantages and disadvantages of each method to support patients’ informed contraceptive decision-making across their reproductive years.
Contraceptive choice is influenced by multiple factors, which can change over an individual’s reproductive life course, from adolescence through to the perimenopause. These include medical eligibility, method effectiveness, risks and side effects, a desire for noncontraceptive benefits for conditions including heavy menstrual bleeding and acne, ease of use, access and costs, as well as personal preference. The role of the practitioner is to support shared and informed decision- making, which includes providing information about currently available methods.
This article provides an overview of long-acting reversible contraceptives (LARCs), combined hormonal methods, progestogen-only pills, depot injections and emergency contraceptive pills. It includes discussion about more recently available options, such as the new smaller-framed lower-dose 19.5 mg levonorgestrel (LNG) intrauterine device (IUD) introduced in 2020, and the drospirenone 4 mg progestogen-only pill available since mid-2021. Other methods, including barrier and permanent methods, are outside the scope of this article. However, it is important to note that the promotion of condom use should be considered in addition to another effective method of contraception for people of all ages who are at risk of sexually transmitted infections (STIs).
The contraceptive consultation
A key component of the contraceptive consultation is ensuring the contraceptive method recommended is safe for the patient to use. In Australia, safety considerations regarding contraception options are guided by the UK Faculty of Sexual and Reproductive Health Medical Eligibility Criteria (UK MEC).1 This system has four categories, outlined in Table 1, which support the practitioner in determining whether it is safe to provide a particular contraceptive method based on the patient’s individual circumstances and medical history.
Although measurement of blood pressure and body mass index (BMI) are not required before initiating nonhormonal methods and most progestogen-only methods, a contraceptive consultation can be an opportune time to perform these routine health checks, as well as checking STI screening needs and whether the patient’s cervical screening test is up to date.
After a method has been selected, practical considerations need to be discussed, including timing of initiation, how to manage method deviations, such as missed pills or late vaginal ring insertion, and practical advice about the management of troublesome side effects, including irregular bleeding patterns. The updated Therapeutic Guidelines provides practical guidance for practitioners on all aspects of contraception and selected patient information sheets.2 The Family Planning Alliance Australia (FPAA) contraceptive efficacy chart and contraception fact sheets can be used to support informed choice and effective method use and are available from family planning organisation websites (www.familyplanningallianceaustralia.org.au/resources/).
Long-acting reversible contraceptives
LARCs comprise hormonal and copper (nonhormonal) IUDs and the etonogestrel (ENG) contraceptive implant. Uptake of IUDs is increasing in Australia, particularly among those aged under 25 years.3 Hormonal and copper IUDs have the highest continuation rates of all reversible methods, with 70% of users continuing to three years of use.4 Pregnancy must be excluded before IUD insertion because of the potential for premature delivery and septic abortion if a pre-existing pregnancy continues and the IUD cannot be removed.5 Screening for STIs in at-risk individuals, such as those aged under 30 years, is recommended before IUD insertion. If a copper IUD is being inserted for emergency contraception within five days of unprotected intercourse, an early pregnancy not yet diagnosable by a urine pregnancy test must also be excluded through the patient’s history about recent contraception use.
Copper IUDs are immediately effective, regardless of which stage of the menstrual cycle they are inserted, but hormonal IUDs are immediately effective only if started between days 1 and 5 of the menstrual cycle and take seven days to become effective if inserted at any other time.
Benefits
IUDs and implants are the most effective LARC methods.6 There are no medication interactions to consider when recommending an IUD.7 They are safe to insert in nulliparous people and in those who have not had vaginal sex, although insertion may be technically more difficult in these circumstances. They have few contraindications, are safe to use during lactation and have high user satisfaction and continuation rates. Both IUDs and implants are rapidly reversible, with an expected quick return to fertility.8 LARCs are also cost effective because of their long duration of use and are a first-line method for people of all reproductive ages.9
Contraindications and complications
Contraindications to IUDs include a current pelvic infection and significant distortion of the uterine cavity. IUDs can be inserted at the time of a surgical abortion and in the postpartum period. Postpartum insertion, including after caesarean section, is UK MEC 1 if performed within 48 hours after delivery, or from four weeks postpartum onwards; however, the insertion of an IUD within 48 hours after delivery is associated with higher expulsion rates than insertion at four to six weeks postpartum. Insertion between 48 hours and four weeks postpartum is UK MEC 3.¹ Because of the increased risk of uterine perforation in the early postpartum period, IUD insertion in the primary care setting is usually delayed until six to eight weeks after a delivery.1,10 Hormonal IUDs have additional contraindications to consider, which are similar to those of other progestogen-only methods (Table 2). Copper IUDs should not be used in those with Wilson’s disease.
Complications associated with IUDs include a risk of perforation, particularly in the postpartum period, and infection in the first 20 days after insertion, with each of these complications occurring in around 0.2% of insertions. The risk of expulsion is around 5%.11 A pregnancy that occurs with an IUD in place has a greater chance of being ectopic, although the overall risk of an ectopic pregnancy is lower than for those not using contraception. Ectopic pregnancies occurring with a hormonal or copper IUD in place account for around 50% and 20% of pregnancies, respectively.12
Hormonal IUDs
The 19.5 mg and 52 mg LNG IUDs are subsidised on the PBS. Their efficacy is 99.7 to 99.9%.12,13 Both are indicated for contraception and the 52 mg LNG IUD is also indicated for the treatment of heavy menstrual bleeding and as part of menopausal hormone therapy to protect the endometrium. Although preliminary studies indicate the 52 mg LNG IUD might be effective as emergency contraception, its use is not currently recommended for this indication.14,15
Both LNG IUDs are licensed for five years’ use, and the 52 mg LNG IUD can be left in place until menopause is determined or until the age of 55 years if inserted in a person aged 45 years or older (off label).11 The 19.5 mg LNG IUD is smaller than the 52 mg LNG IUD, appears to be easier to insert and is associated with less pain on insertion. In a community study, around two-thirds of nulliparous users reported their insertion pain as none or mild.16
Benefits
Although both LNG IUD types have been shown to decrease bleeding days and dysmenorrhoea, only the 52 mg LNG IUD has been extensively studied for these benefits.17 It has been shown to be effective in reducing endometriosis and adenomyosis-associated dysmenorrhoea and is very effective at reducing heavy menstrual bleeding.18-22
Side effects
Irregular bleeding or spotting in the first few months of use is common with both devices.17 The 19.5 mg LNG IUD appears to be associated with a higher mean number of bleeding or spotting days and lower rates of amenorrhoea than the 52 mg LNG IUD.23 However, the significance of these differences to users may be small as discontinuation of either IUD because of bleeding problems is uncommon.24,25
Although the 19.5 mg LNG IUD is associated with around a 50% lower systemic exposure to LNG than the 52 mg device, initial data do not show any significant differences in acne, breast discomfort, pelvic pain or weight gain between devices.17 Both devices increase the risk of benign, asymptomatic ovarian cysts, with the 52 mg LNG IUD having a higher risk. Cysts are likely to resolve spontaneously.17
Copper IUDs
Copper IUDs are around 99.5% effective, have the advantage of being hormone-free and provide extremely effective emergency contraception.12,26 Two copper IUDs are available in Australia, lasting five or 10 years, depending on the type. Both can be left in place until menopause if inserted at the age of 40 years or over.11 They are not subsidised on the PBS and cost around $75.
Use of copper IUDs is associated with heavier and slightly more prolonged menstrual bleeding and has no benefit in relation to dysmenorrhoea.19,27 These factors are an important consideration in choice of IUD type (copper or hormonal) for those with heavy menstrual bleeding, iron deficiency anaemia and endometriosis. Irregular bleeding is common in the first few months of use but usually settles.
Etonogestrel implant
The ENG implant is PBS subsidised. It lasts for three years, regardless of the user’s BMI, and is 99.95% effective.28,29 The ENG implant can be started any time postpartum, including in those who are breastfeeding, and can continue to be safely changed three-yearly until menopause is determined or until the age of 55 years. Unlike IUDs, its efficacy can be affected by the concurrent use of liver enzyme-inducing medications and switching to an alternative unaffected method is recommended for long-term use (Table 3).7 Major contraindications are as for other progestogen-only methods (Table 2).1
Starting an ENG implant
The ENG implant can be inserted anytime in the menstrual cycle. It is immediately effective if started between days 1 to 5 of the cycle; otherwise, it takes seven days to become effective. When started at a time that an early undetectable pregnancy cannot be excluded (referred to as ‘Quick Start’), a urine pregnancy test is recommended in four weeks’ time, with a follow-up system in place to ensure a possible pregnancy diagnosis is not delayed.30 An ENG implant can be inserted straight after a delivery or surgical abortion, or within the first five days of a medical abortion, including at the time of prescribing mifepristone (the first tablet of a two-step process).31 It cannot be used to protect the endometrium as part of menopausal hormone therapy.
Complications and side effects
Insertion and removal of the ENG implant may cause scarring and a local reaction and carries a small risk of infection. Deep insertion may sometimes occur, and specialist intervention is required for removal of all impalpable devices. It is now recommended that the device be inserted over the triceps, to avoid the rare complication of intravascular insertion.32,33
Troublesome bleeding is the most common side effect of the ENG implant. It is important to inform users upfront that their bleeding pattern will change but that it is not possible to predict on an individual basis how it will change. Amenorrhoea occurs in around 20% of users and frequent bleeding (more than five episodes per 90 days) or prolonged bleeding (an episode that lasts more than 14 days) in 25%. Around 55% have no or light and infrequent bleeding. Generally, the initial bleeding pattern predicts ongoing bleeding, although there is some settling during the first three to six months after insertion.34 Users should also be provided at the time of insertion with information about the availability of strategies for managing troublesome bleeding should it occur (Box).
Users may also experience headaches, mood changes or emotional lability, acne, breast tenderness and loss of libido.35 Pre-existing acne improves in some users.36 Weight gain is a frequently reported side effect, although causal evidence is lacking.35-38
Combined hormonal contraception
Combined hormonal contraceptives (CHCs) include combined oral contraceptives (COCs) and the vaginal ring. COCs are the most common method of contraception used in Australia with a large number of choices available, containing ethinylestradiol, estradiol or its pro-drug estradiol valerate in combination with one of nine progestogens.39 The vaginal ring is a slow-release combination of ethinylestradiol and ENG and is treated in the same way as COCs with regard to drug interactions, side effects and contraindications. It is not PBS listed. CHCs are safe to use in those without contraindications until the age of 50 years, after which switching to another method is recommended. They are generally considered safe to use from six weeks postpartum onwards, including in those who are breastfeeding. CHCs are 99.5% effective when used perfectly, but only 93% effective during the first year of use in typical use, usually because of missed pills or delayed vaginal ring insertion.40
Benefits
CHCs are associated with a decrease in acne, menstrual pain and bleeding. Users can manipulate their menstrual cycles.41 CHCs also decrease the risk of ovarian, endometrial and bowel cancer, and can have benefit in managing premenstrual dysphoric disorder and perimenopausal symptoms.42-44
Contraindications
Contraindications for CHCs are mostly related to risk factors for, or a personal history of, arterial and venous disease. MEC 3 and 4 contraindications include a history of migraine with aura, smoking in those aged over 35 years, hypertension, BMI above 35 kg/m2, a first-degree relative aged 45 years or younger with a venous thromboembolism (VTE), or a personal history of breast cancer. Table 4 summarises the important MEC 3 and 4 conditions for CHC use. CHCs are not generally recommended for those concurrently using a liver enzyme-inducing medication (Table 3).7
Starting a CHC
CHCs can be started at any time of the cycle and will be immediately effective if active hormones are started on days 1 to 5 of a normal menstrual cycle. Otherwise, they take seven days to become effective.
When starting a COC, some pills are packaged to start with a nonhormonal sugar pill and will take longer to be effective. If an early undiagnosable pregnancy cannot be excluded, a pregnancy test is recommended in four weeks’ time, even if scheduled bleeding occurs.45
Active hormone pills can be taken continuously for extended periods to minimise bleeding by running pill packets or vaginal rings together without a hormone-free break. A hormone-free break of four days can be initiated if unscheduled bleeding occurs, as long as at least 21 active pills have been taken consecutively before the break.46,47 There is one extended use COC available, with a 30 mcg ethinylestradiol and 150 mcg LNG combination pill packaged for the user to take 84 consecutive hormone pills, followed by seven 10 mcg ethinylestradiol pills. It is designed for bleeding to occur once every three months, as opposed to once per month as with other COCs.48
Blood pressure and BMI should be documented at initiation and annually. No routine investigations are necessary.45 In those who have no contraindications, prescribing 12 months of CHCs is important to maximise continuation rates.49,50
Choosing a CHC
A low-dose pill containing ethinylestradiol 20 or 30 mcg and LNG is the recommended first-choice CHC.45 These pills have been extensively studied and have similar discontinuation rates to other CHCs when compared in head-to-head trials.51,52 Some brands of these pills are subsidised on the PBS. A list of COCs available in Australia appears on the Family Planning Victoria website (www.fpv.org.au/assets/resources/Combined_hormonal_contraceptive_pill_chart_UPDATED_11.21-005.pdf). Any other CHC can be used first line, but there appears to be a small increased risk of VTE with ethinylestradiol pills containing cyproterone acetate, desogestrel, dienogest, drospirenone or gestodene and the vaginal ring compared with pills containing ethinylestradiol 20 to 30 mcg and LNG or norethisterone.53
Risks
Serious risks
Some serious risks are associated with CHC use; however, the absolute risk for most individuals of reproductive age is low. All CHCs increase the risk of VTE to around threefold above baseline, with the highest risk occurring in the first year of use.54,55 There appears to be a small increase in ischaemic heart disease and stroke.56 There may also be a small but reversible increased in risk of breast cancer.57 Although the use of CHCs is associated with a small increase in the risk of cervical cancer, the attributable risk is likely to be low in Australia because of the human papillomavirus vaccination and national cervical screening programs.42
Other risks
Use of CHCs is associated with a small increase in blood pressure, with the exception of drospirenone-containing pills and pills with estradiol or estradiol valerate in place of ethinylestradiol.58-60 There is a small increase in the risk of inflammatory bowel disease.61 Although evidence is insufficient to determine whether there is an increased risk of gallbladder disease, there are some restrictions in the use of CHCs in those with the condition.62
Several side effects have been reported by users of CHCs; however, it is difficult to prove cause and effect. Weight gain is a common complaint but has not been demonstrated for pills containing 35 mcg or less ethinylestradiol.63 Unscheduled bleeding is common, particularly with the lowest ethinylestradiol dose pills, but usually settles over a few months.64 General side effects of CHCs often settle with time and include:65
- headache
- nausea
- breast tenderness
- unscheduled bleeding
- amenorrhoea
- acne (usually improves)
- bloating
- mood changes
- reduced libido
- weight gain
- melasma (also known as chloasma).
- In addition, vaginal ring users may report:
- increased vaginal discharge
- device discomfort for the user or partner
- expulsion of the ring.
Missed CHC pills
A COC pill is not considered missed until it is more than 24 hours late. The rules for missed COC pills can also be applied to the vaginal ring and are outlined in the Flowchart.
Progestogen-only pills
Two types of progestogen-only pills (POPs) are available in Australia, which differ in their mechanism of action. The ‘traditional’ low-dose pills containing LNG 30 mcg or norethisterone 350 mcg have been available for many years and are PBS-listed. They act primarily by thickening the cervical mucus and have a narrow three-hour window of pill taking to maintain efficacy.66 Recently, a new POP type containing 24 tablets of drospirenone 4 mg and four inactive sugar tablets has become available that primarily acts to suppress ovulation and has a 24-hour window for tablet taking.67 The drospirenone 4mg POP is not available on the PBS.
The efficacy of both types of POPs is considered the same as that of CHCs: 99.5% in perfect use and 93% in typical use.40 However, traditional LNG- and norethisterone-containing POPs are considered to have a more vulnerable efficacy, and failure rates are higher in those aged under 40 years than in older users.68 Both types of POPs can be used until menopause is determined or until 55 years of age and are safe to use at any time postpartum, including in those who are breastfeeding.
Benefits, contraindications and medication interactions
A small study in adolescents using the drospirenone 4 mg POP showed a reduction in dysmenorrhoea and use of pain medication to control it.69 POPs provide a safe alternative in those with contraindications to CHC, such as migraine with aura, and are an appealing alternative to LARCs in those who do not wish to undergo a procedure.
Major contraindications are as for other progestogen-only methods (Table 2).1 Efficacy can be affected by the concurrent use of liver enzyme-inducing medications, and the POP is not a recommended in long-term users of these medications (Table 3).7
Starting a progestogen-only pill
POPs can be started at any time in the cycle and will be immediately effective if started on days 1 to 5 of the menstrual cycle. Otherwise, the traditional POPs take 48 hours (three pills) and the drospirenone POP takes seven days to become effective. If an early undiagnosable pregnancy cannot be excluded, a urine pregnancy test is recommended in four weeks’ time, even if scheduled bleeding occurs.45
Side effects and serious risks
To date, no serious risks have been established for the POP. The most common side effect is irregular bleeding, which occurs in around 40% of those taking traditional POPs; around 20% experience amenorrhoea, and 40% have regular cycles.70-72 The drospirenone 4 mg POP has been developed to induce a monthly scheduled withdrawal bleed during the hormone-free break. Although unscheduled bleeding is initially common, both scheduled and unscheduled bleeding and spotting decline over time. Amenorrhoea increases from 10% in the first cycle to 45% in cycle 9.73 Overall bleeding patterns are generally acceptable to users.74,75 Other side effects can include acne, mood changes, weight gain and loss of libido.
Missed POPs
A traditional POP is considered to be a missed pill when it is taken three or more hours late. Condoms should be used during intercourse until three consecutive pills have been taken, and emergency contraception should be considered if unprotected sex occurs during this time. The missed pill rules for the drospirenone 4 mg POP are the same as those for CHCs (Flowchart).
Depot medroxyprogesterone injection
Depot medroxyprogesterone acetate (DMPA) is PBS subsidised and is given by deep intramuscular injection 12 weekly (± 2 weeks). It is 99.8% effective in perfect use and 96% in typical use.40 Its efficacy is unaffected by liver enzyme-inducing medications and it is generally considered safe to use at any time postpartum or postabortion. Because of theoretical concerns about effects on lipids and bone density, a switch to another method is recommended at the age of 50 years.76
Starting DMPA
DMPA can be given any time in the menstrual cycle. However, if an early undetected pregnancy cannot be excluded, a pregnancy test is recommended in 4 weeks’ time, with a follow-up recall ystem in place. It can be given straight after a delivery or a surgical abortion, or within the first five days of a medical abortion, including at the time of prescribing mifepristone (first tablet of a two-step process). A concern is that administration around the time of mifepristone may be associated with a small increase in the risk of a failed abortion.31
A blood pressure check is required at initiation of DMPA and on annual assessment. Measurement of lipid levels is recommended for those with cardiovascular risk factors who are considering this method. Baseline BMI can be useful to measure as DMPA use has been associated with weight gain.77
Contraindications, side effects and serious risks
Major contraindications are similar to other progestogen-only methods, with additional precautions related to cardiovascular disease risk (Table 2).1
Troublesome bleeding is the most common side effect of DMPA, although at least 50% of users become amenorrhoeic. Users should be informed that their bleeding pattern will change but that it is not possible to predict how it will change on an individual basis. Users should also be provided with information at the time of insertion about the availability of strategies for managing troublesome bleeding should it occur (see Box).
Unlike other methods of contraception, DMPA is associated with weight gain. Around 20% of users gain 10% of their body weight in the first year of use.77 Use of DMPA is associated with loss of bone density that is considered reversible.78-81 Data on the effects on fracture risk are inconclusive.82,83 Detailed assessment of risk factors for osteoporosis should take place for new users, particularly those aged under 18 years, and every two years for continuing users.45 Users also may complain of headaches, mood changes including emotional lability, acne, breast tenderness and loss of libido.
Emergency contraception
The most effective emergency contraception is a copper IUD inserted within five days of unprotected intercourse.26 It also provides ongoing long-term contraception, although finding a provider who can insert an IUD within the required timeframe can be challenging.84
Two types of emergency contraceptive pills (ECPs) are available over the counter at pharmacies; the ulipristal 30 mg ECP and the LNG 1.5 mg ECP. Both act by preventing or delaying ovulation and prevent around 85% of expected pregnancies.85 Both are safe to use, with no known contraindications except allergy to the constituents and a known pregnancy. Should they be inadvertently taken in early pregnancy, there is no evidence of a teratogenic effect on the fetus. Ulipristal is licensed for use up to five days (120 hours) after unprotected intercourse and, although its efficacy is maintained across this period, users are advised to take it as early as possible for an optimal effect. LNG ECPs are licensed up to three days (72 hours) but have some effectiveness up to 96 hours after unprotected intercourse.86 Emergency contraception is not subsidised on the PBS. See Table 5 for a comparison between ECP types.1,2,85-91
Conclusion
Raising awareness about all contraceptive options is important for facilitating informed choice. The method that suits an individual during adolescence may be very different to the method that meets their needs during the mid-reproductive years and later at the perimenopause. Efficacy, contraindications, adverse effects and potential benefits of each contraceptive option should be discussed to help find a suitable method for the individual patient. MT
